Characterization of the mechanisms of daptomycin resistance among Gram-positive bacterial pathogens by multidimensional lipidomics
Characterization of the mechanisms of daptomycin resistance among Gram-positive bacterial pathogens by multidimensional lipidomics
Abstract
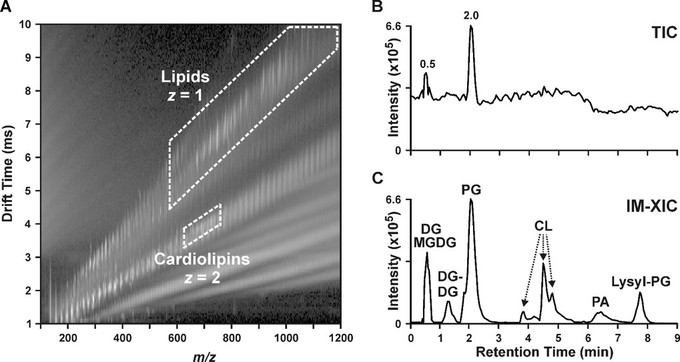
Previous work suggests that altered lipid metabolism may be associated with daptomycin resistance in Gram-positive pathogens, but lipidomic changes underlying resistance are not fully understood. We performed untargeted lipidomics by using three-dimensional hydrophilic interaction liquid chromatography-ion mobility-mass spectrometry (HILIC-IM-MS) to characterize alterations in the lipidomes of daptomycin-susceptible and -resistant isogenic strain pairs of Enterococcus faecalis, Staphylococcus aureus, and Corynebacterium striatum. We first validated the HILIC-IM-MS method by replicating the expected alterations of phospholipid metabolism in the previously studied E. faecalis strain pairs, such as reduced phosphatidylglycerols (PGs), while also revealing additional changes in cardiolipins (CLs), lysyl-PGs, and glycolipids. Whole-genome sequencing of the S. aureus and C. striatum strains found that daptomycin resistance was associated with mutations in pgsA, which encodes phosphatidylglycerophosphate synthase, as well as mutations in genes affecting fatty acid biosynthesis and cell wall metabolism. Lipidomics revealed significantly decreased levels of PGs, CLs, and amino acid-modified PGs, as well as accumulation of lipids upstream of PGs, such as glycolipids and phosphatidic acids, in the resistant strains. Notably, the glycolipids, diglucosyldiacylglycerols, were significantly elevated in a fatty acid-dependent manner in the daptomycin-resistant S. aureus strain. In daptomycin-resistant C. striatum, which has a unique cell envelope architecture, the glycolipids, glucuronosyldiacylglycerols, and phosphatidylinositols were significantly elevated. These results demonstrate that alteration of lipid metabolism via mutations in pgsA is a common mechanism of daptomycin resistance in two distinct species of Gram-positive bacteria and point to the potential contribution of altered glycolipid and fatty acid compositions to daptomycin resistance.